
Andrey Solver, February 14, 2025.

Elon Musk and Peter Diamandis talk about the largest prize in history for a method and technology for CO2 removing.
It takes place in Florida on April 22, 2021.
I don't know the exact location in Florida where this video was filmed, but I know exactly what is under the feet of these gentlemen.
At a depth of 42 feet (12.8 meters) there is limestone, CaCO3 or calcium carbonate.
The solution to the main global problem is literally under their feet.
Let's get to the bottom of it.
![]()
Florida has repeatedly been the bottom of the ocean. For example, during the time of the Greenhouse Earth 100-80 million years ago, the place where people are now talking about carbon dioxide was under water. About 300 meters below the surface of the ocean, the level of which was high because there were no polar ice caps. There was 10 times more CO2 in the atmosphere – about 3,900 ppm. The Earth was going through a period of high volcanic activity, the American continents broke away from Africa and drifted away, opening and expanding the Atlantic Ocean
The ocean level fluctuated, the land appeared and disappeared under the sea water. During the Miocene period, the sea level rose 22 million years ago, 17 and 10 million years ago to 300 feet higher than today. The slow movement of the Earth's crust and the water level formed the processes of deposition of marine sediments in shallow water. Florida limestones are deposits of calcite-producing marine organisms: corals, shells and unicellular foraminifera. Over millions of years, sediments accumulated several kilometers thick and formed the Florida Carbonate Platform.
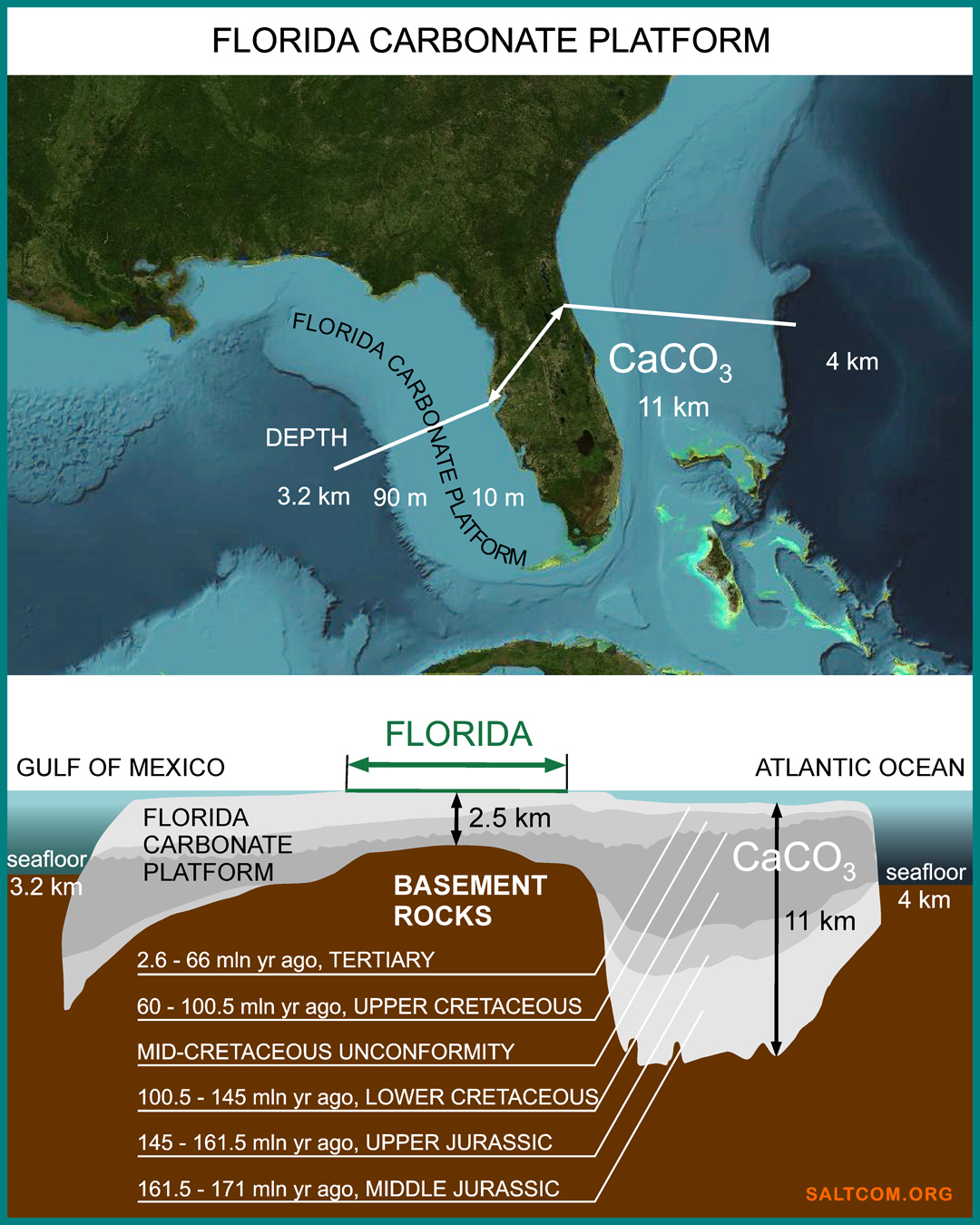
The average height of Florida above sea level is 30 meters, and, for example, Miami is only 4 meters. If we talk about the thickness of non-carbonate rocks or about the soil, then we are talking about centimeters. The blooming peninsula is a thin film of life on top of a huge mountain of limestone, CaCO3, extending 11 kilometers deep. This is the result of the removal of atmospheric CO2 over the last 170 million years.
In oceanology, shallow water of platforms are called "carbonate factories" because of the regular production of carbonate mineral sediments.
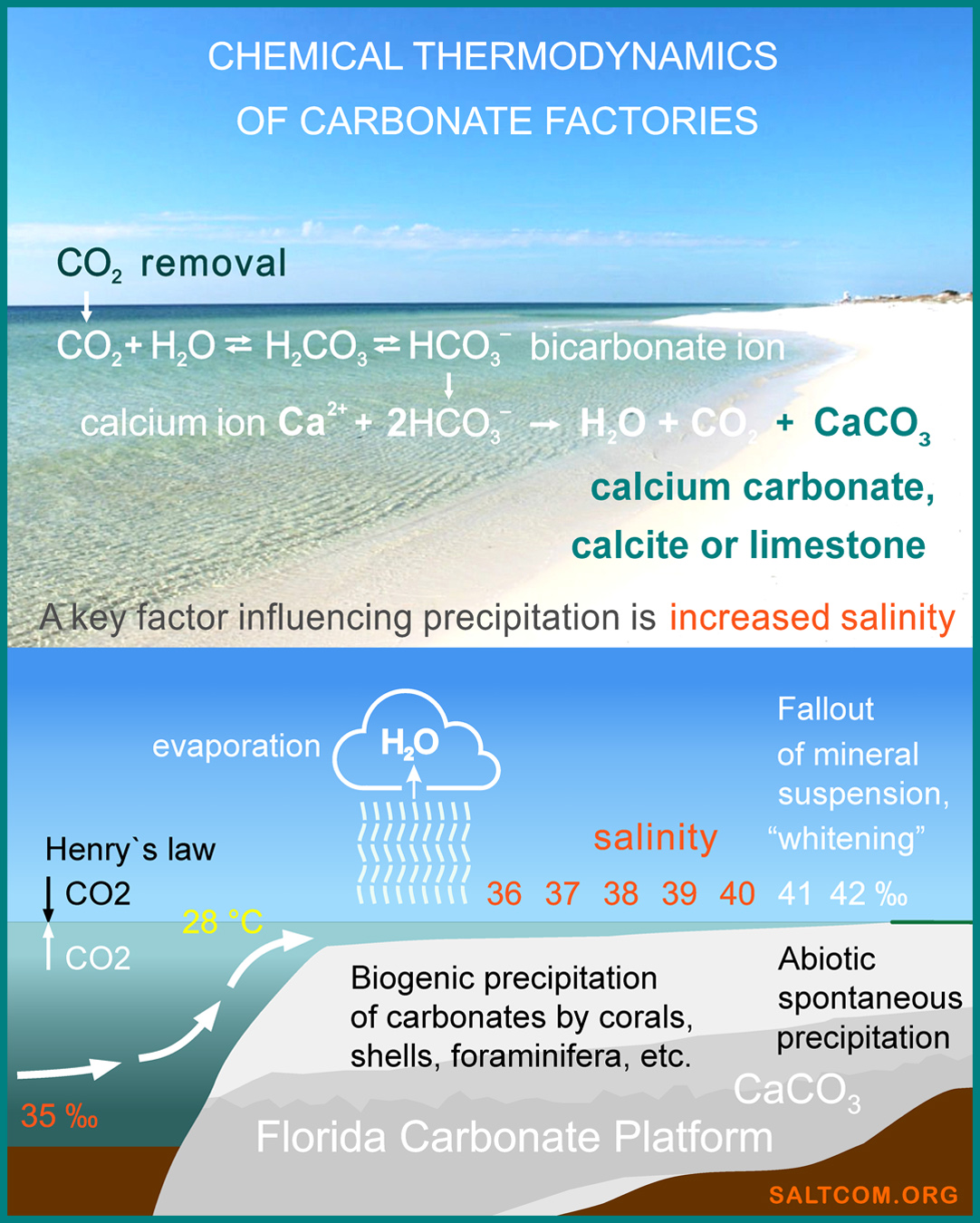
Researches show that in these factories, the main engine is salt.
At a relatively constant surface temperature of about 28°C, due to evaporation, salinity increases from the global average of 35‰, parts per thousand, to 41-43‰. The higher the salinity, the greater the ionic strength of the solution — a measure of the intensity of the electric field created by ions in the solution. According to chemical thermodynamics, it affects the solubility constants of carbonates, causing them to precipitate from seawater. This strength causes marine organisms to bind carbonate ions into insoluble minerals: skeletons, tests and shells. And during a hot period, when the salinity exceeds 41‰, direct chemical precipitation of aragonite, a polymorph of CaCO3 , is possible, which then recrystallizes into calcite. This phenomenon is called "whitening" — the spontaneous abiotic formation of mineral suspension in seawater.
The productivity of local carbonate factories is very high. Thus, the precipitation of carbonates on the neighboring Bahamian platform averages 0.6 kg per square meter per year, and at the edge of the platform near the Strait of Florida reaches 1.2 kg/m2/year CaCO3 or, when calculated by weight of carbon dioxide, 0.5 kg CO2/m2/year.
This carbon pump is capable of pumping out all the CO2 from the atmospheric column above it (0.4 kg/m2) in less than 1 year.
These are real "white holes" that absorb carbon from the atmosphere and ocean.

Since salt initiates and accelerates the precipitation of carbonate ions from seawater, its deficiency accordingly slows down the work of carbonate factories. Global warming, melting of glaciers and polar caps lead to desalination of surface waters. The thermodynamic coupling of salinity and pH level of water leads to the so-called "ocean acidification", a decrease in foraminifera productivity and coral bleaching.
This happens on the surface. At the bottom of the Gulf of Mexico, the picture is completely different: there are bottom supersaline lakes with salinity 7-8 times higher than the average. Under the bottom limestones, there are layers of strong brines, and even deeper – massive layers of rock salt several kilometers thick.
To get to the bottom of it, let's look deeper into time, to inception.
Tectonic movements of the earth's crust have formed gigantic salt deposits in the Mexican salt basin, the second largest in the world.
To view the global picture of salt, its deposits and mining, please refer to the Salt of the Earth.
![]()
Rock salt has a lower density (2.2 ton/m3) than the rocks above it (2.6 – 2.8 ton/m3), at a depth of several kilometers and at high pressures it becomes very fluid, penetrating through the overlying layers, forms diapirs and salt domes.
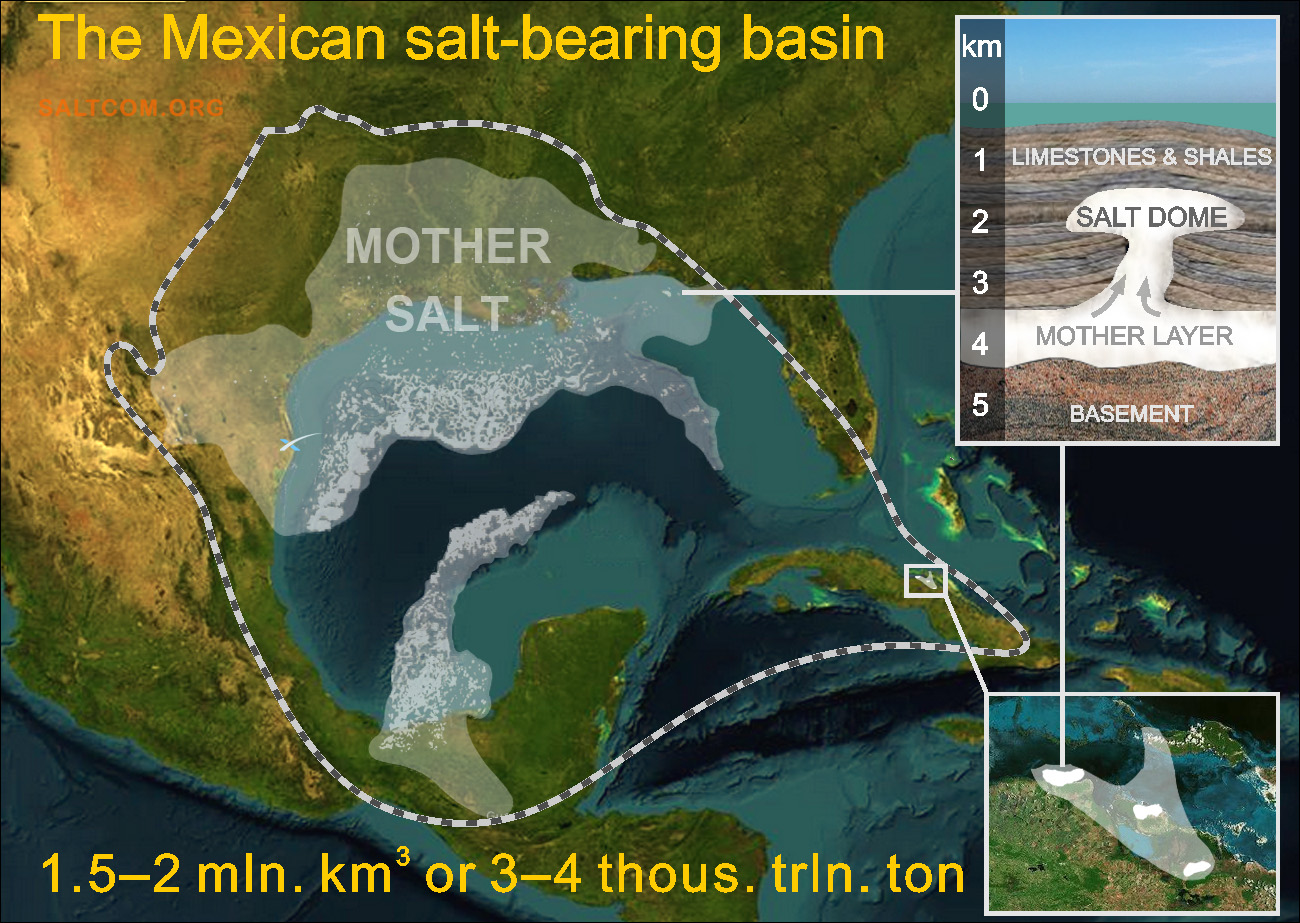
The mother salt layer is an almost continuous coating with a thickness of about 1-1.5 km, from which diapirs and salt domes grow to the surface at a rate of 1-2 mm per year. The area of these salt bodies ranges from several to several tens of kilometers. Vertical dimensions: 3-5 km of salt in the continental part and up to 10 km in the deep-water areas of the golf.
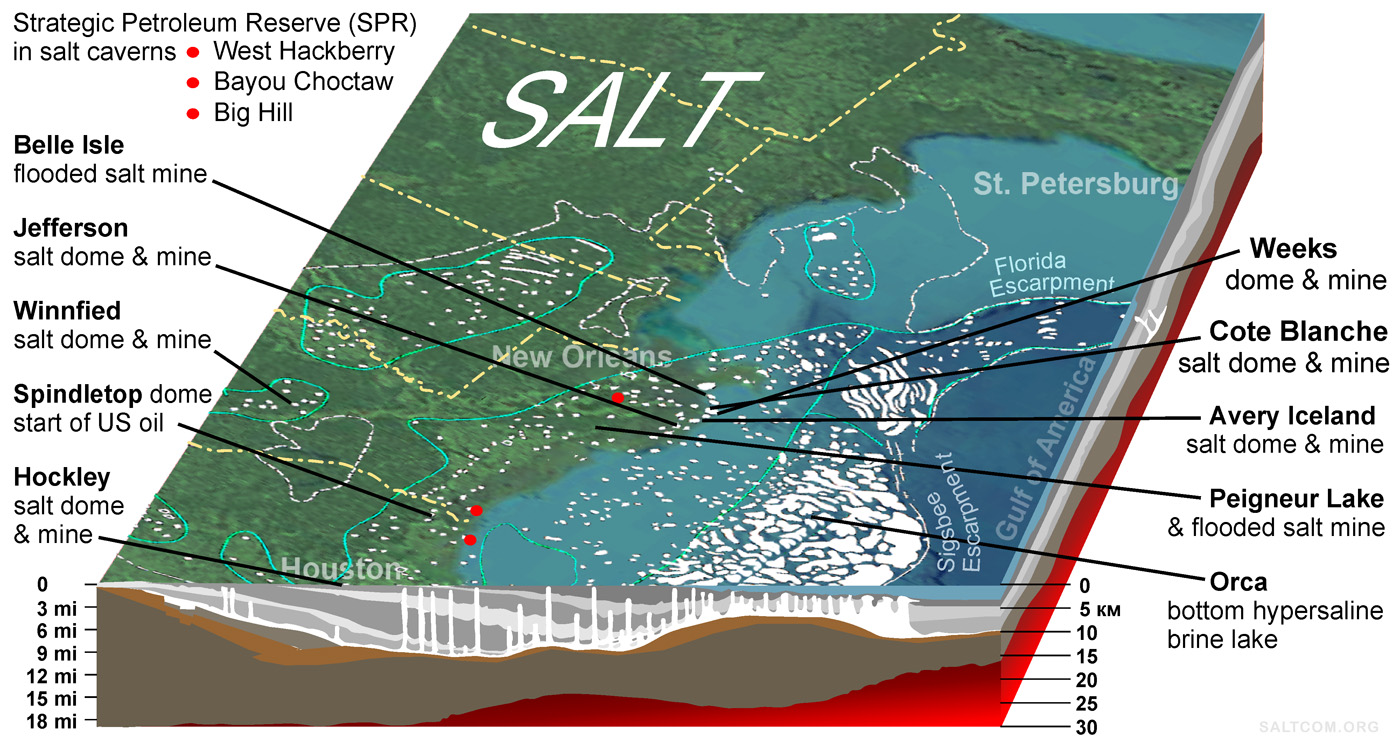
Most salt bodies are located at a depth of more than 2 km, but there are salt domes that come to the surface. In the continental western part of the state of Florida, there are two of domes 250 m from the surface, but in Mississippi, Louisiana and Texas there are several domes that reach the day surface.
The salt mining industry in these regions of the US is about 10 million tons of rock salt per year.
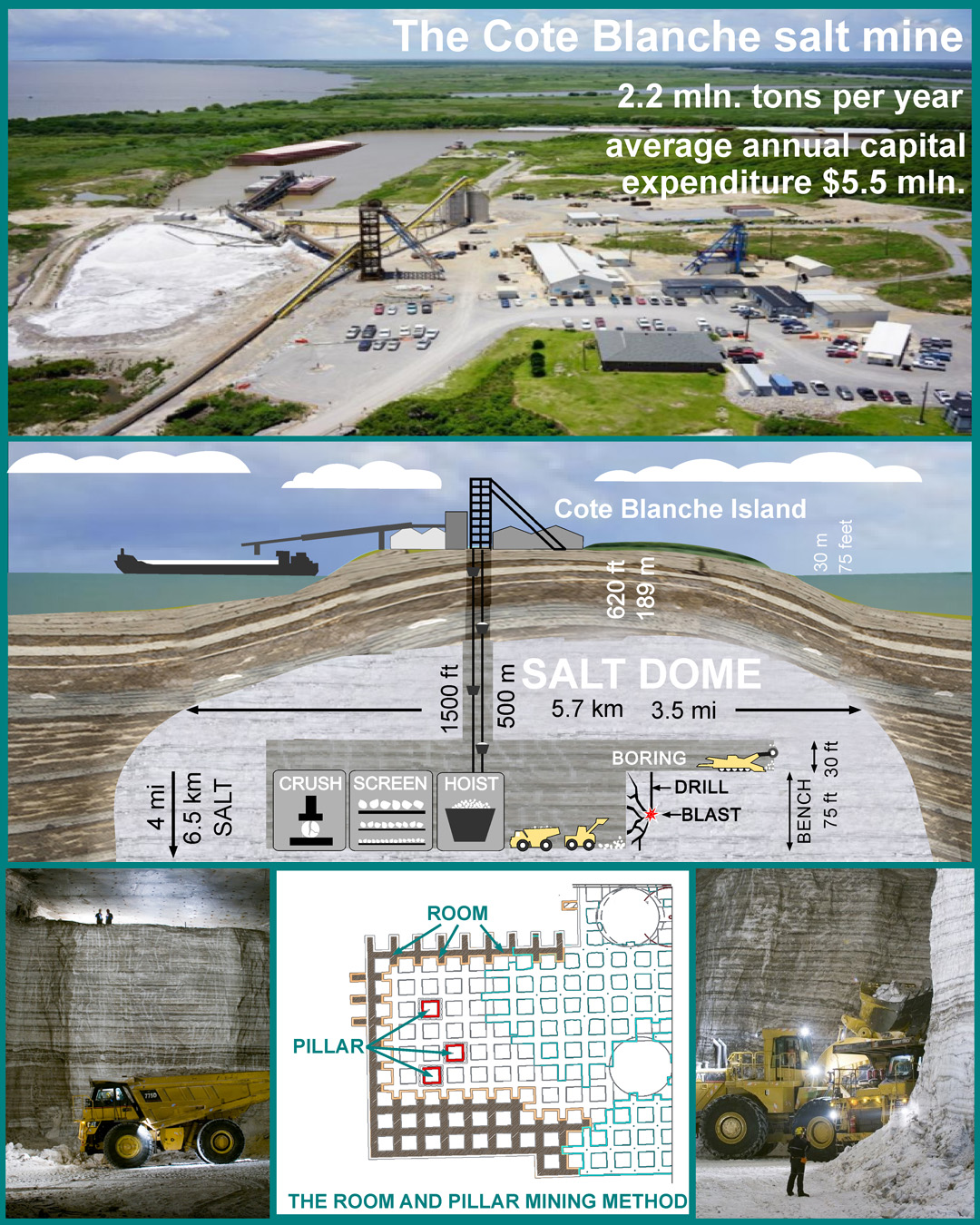
Rock salt is mainly used for road deicing. The final price in the US market is $40-60 per ton.
Salt for animal husbandry, chemical and food industries is extracted mainly by liquid method – brine-mining, by drilling wells, pumping water into them, pumping out brine, and evaporation both in outdoor pools and in vacuum pans. Worldwide, brine-mining can exceed the mining of dry salt.
The technologies for creating and operating underground salt caverns for storing oil and gas are also well developed. The US Strategic Petroleum Reserve is located in such underground storage facilities in salt domes on the Gulf coast.
Also for our purposes, several flooded salt mines can be used as salt sources. Over the years, the brine has reached a maximum salt concentration of 35.9% in water, and with proper operation, these mines can become a long-term source of brine with 1/3 of the salt.
![]()
The illustration shows a scheme of direct chemical removing of carbonate ions. At a temperature of about 28°C, a critical excess of the ionic strength immediately leads to solid precipitation. However, milder and longer-term metods may also be feasible, focused on biological catalysts (carbonic anhydrase) of the calcite–fixing microbiota (foraminifera and coccolithophorides, the main carbon removers on Earth).
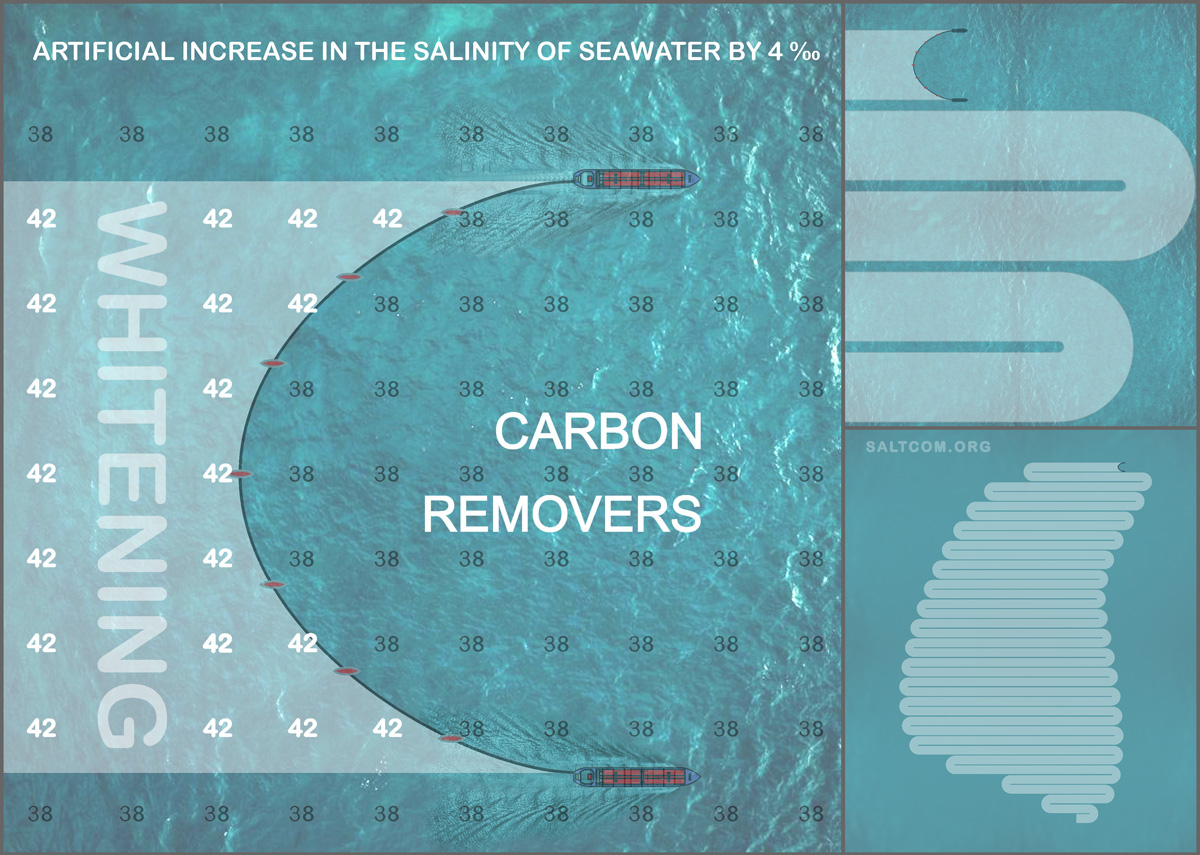
In order to raise the salinity of 2 meters of surface seawater by 4 ‰ and cause mineral precipitation, 8000 tons of salt per 1 square kilometer are needed. For example, two medium-sized bulkers (Handymax) with a capacity of 50,000 tons each will be able to remove dissolved CO2 on an area of 12.5 km2.
The total mass of the deposited CaCO3 depends on many factors of a specific location and can be calculated by mathematical modeling of chemical kinetics over time. For direct "whitening", the time is seconds and minutes, and for the intensification of "foraminiferal calcite rain" – months and years. According to the "Bahamian model", 1 km2 produces on average about 600 tons of CaCO3 per year.
It should be understood that salt does not disappear anywhere and continues its work during diffusion. Thermodynamic calculations show that CCPP, Calcium Сarbonate Precipitation Potential, does not depend on the degree of dissolution. No matter how big the world's oceans and small impacts are, everything amounts to measurable quantities. The resulting factor for the ratio salt/CO2 is the time. In the presented scheme, equivalence occurs after 8000/(600x0.44 molar mass CO2) = 30.3 years.
![]()
Salt is the cause of oil. The complete impermeability of salt formations creates traps for oil and gas, and fluidity at high pressures creates breaks in the layers, tectonic shifts that promote the movement of organic matter and the formation of underground oil reservoirs.
Of the more than 6,000 oil wells drilled offshore in the Gulf of America, most have encountered salt. They can be considered sources of strong brines.

Today, the cost of mining rock salt in the world is in the range of $10-30 per ton. The cost of salt in the form of brine has not been determined due to the lack of demand and practical use. Whereas at a cost of $80 per barrel, 1 ton of crude oil costs about $575 and approximately 3.67 tons of CO2 are generated during its combustion.
Over a thirty-year horizon, the cost of salt for its disposal will be $20 x 3.67 = $73.4 or about 12.7%, which, in my opinion, is quite acceptable for an "indulgence" that offsets CO2 emissions.
![]()
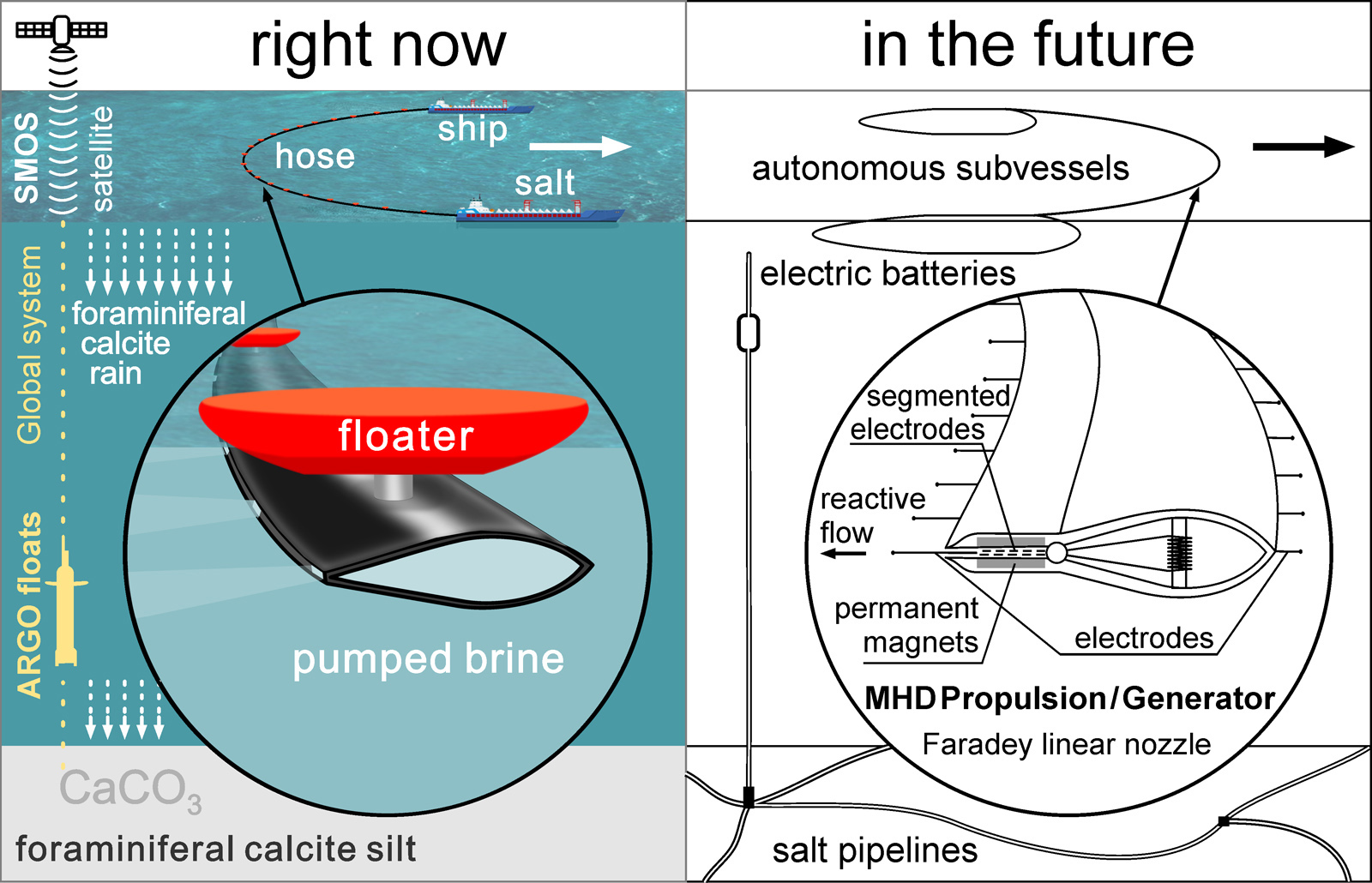
For extract salt it is not necessary to drill wells. In the Gulf of America, at the bottom of the Texas-Louisiana slope, there are several hypersaline lakes, of which the Orca (27°10N 91°30W) is the largest and most studied.
Any ship that can lower a hose to a depth of 2,500 meters can become a salt miner.

With 1/4 salt content in the brine, three quarters of the ballast water will have to be moved. However, this disadvantage can be compensated by the convenient location of the reservoir. The central part of the Gulf of America is a source and intensifier of hurricanes. The fresh water of the Mississippi is lighter than salt water and it spreads over the surface of the Gulf. Whereas salty and warm water comes from the south. The combination of high temperature and low salinity, according to Raoul's law, forms a kind of "boiler" with high evaporation, with the formation of updrafts of moist air that initiate and intensify hurricanes.
Please see how we plan to rid the coast of this natural disaster, how we will cancel hurricanes.
![]()
Where? The deposition of carbonates is influenced by temperature and salinity, with salinity having a greater effect. For direct "whitening", a combination of high temperatures from 28 °C and above (orange-red color) is necessary, and high salinity of more than 37 ‰ – pale-white on map.
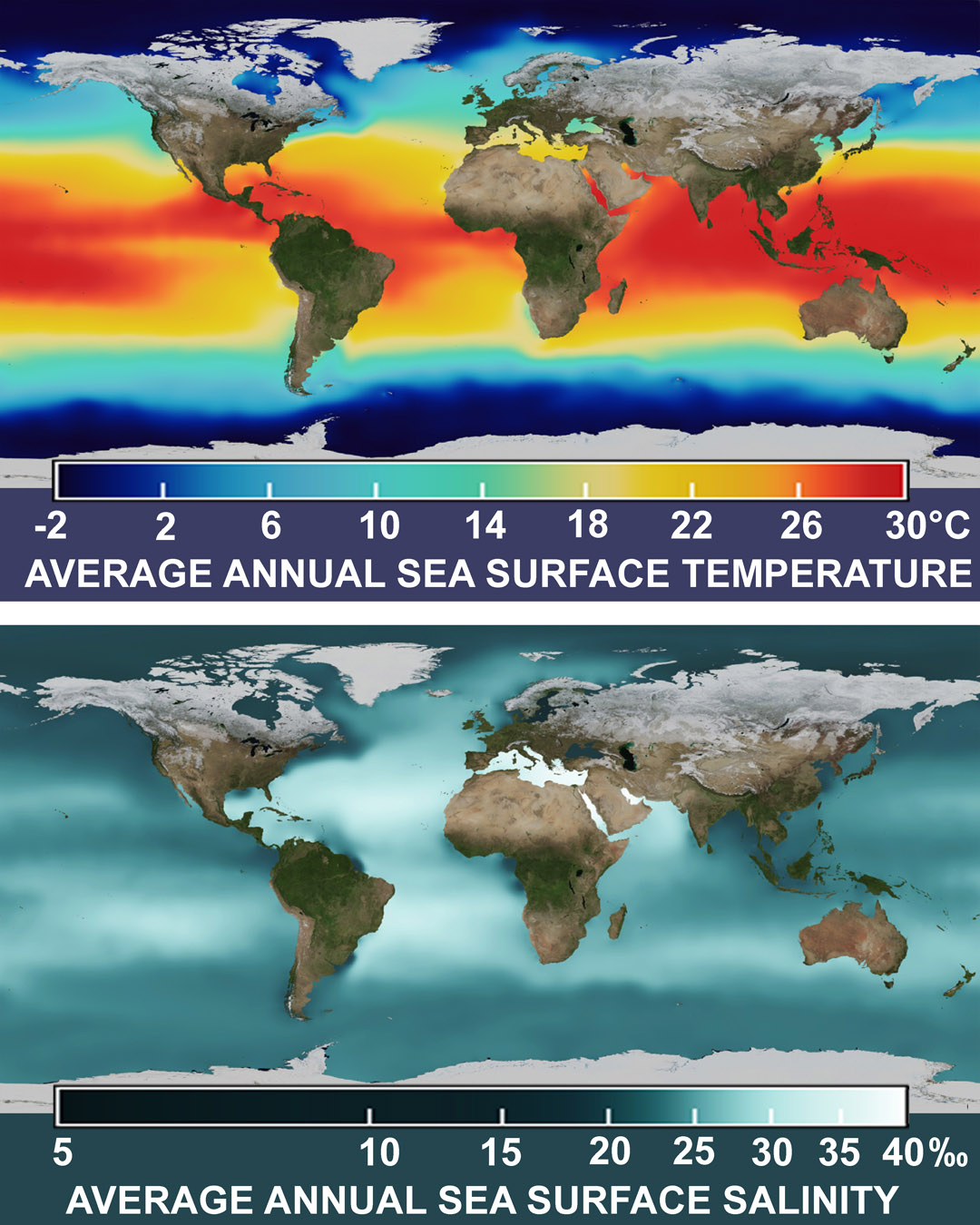
The maps show average values that are higher in summer. The red stripe is shifting upward in the northern hemisphere. And, for example, during the hot season in the Mediterranean Sea, artificial "whitening" is quite possible, taking into account the high salinity of the sea (38 ‰), what is not obvious from the average temperature map (the Mediterranean is only yellow).
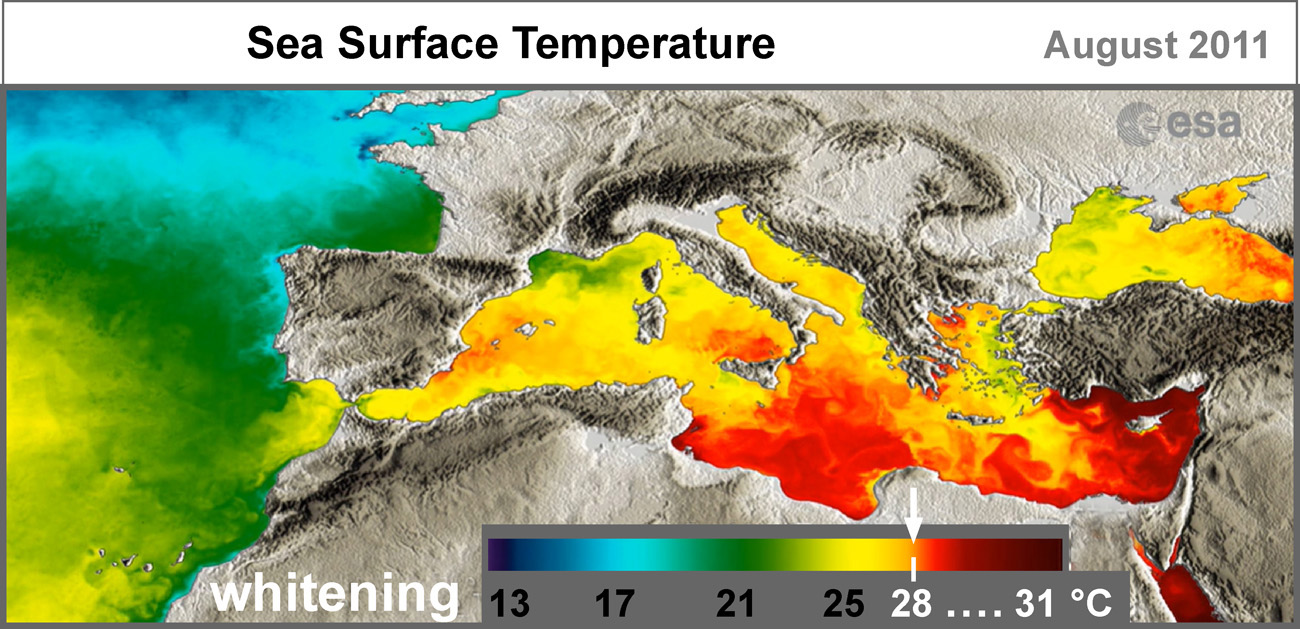
The orange and red colors on the temperature map of the Mediterranean Sea show that "whitening" is possible in summer off the coasts of Spain, Italy, Greece, Turkey, and off the coasts of African and Middle Eastern countries, temperature and salinity values are such that very little exposure is needed to initiate chemical precipitation of carbonates, increasing salinity by only 1 ‰. And in the warm and salty Red Sea and the Persian Gulf, during the hot season, "whitening" happens without any intervention, spontaneously, and their bottom sediments are 95% composed of calcium carbonate, CaCO3, removed carbon.

We are taking part in the final of the XPRIZE Carbon Removal. These are the main goals and rules of this competition.
|
"I'm looking for a hundred million dollar answers that can help change the course of human history and help heal our planet ". |
|
“You have to demonstrate that your methodology is gonna contain the the CO2 in some fashion for 100 years at least”. |
|
|
”Winning team has to prove to our judges that their approach can actually scale to a gigaton level”. |
|
“They've got to actually build something that works and demonstrate something that can extract 1000 tons per year, a kiloton of carbon per year as a demo scale model”. |
P. S. How the competition ended, watch here .